Myriad is focused on three strategic imperatives: transitioning and expanding its hereditary cancer testing markets, diversifying its product portfolio through the introduction of new products and increasing the revenue contribution from international markets. Myriad discovers and commercializes molecular diagnostic tests that: determine the risk of developing disease, accurately diagnose disease, assess the risk of disease progression, and guide treatment decisions across six major medical specialties where molecular diagnostics can significantly improve patient care and lower healthcare costs. is a leading personalized medicine company dedicated to being a trusted advisor transforming patient lives worldwide with pioneering molecular diagnostics. It is estimated that 1.4 million people in the United States and Europe who are diagnosed with cancers annually may be candidates for treatment with DNA-damaging agents. In previously published data, Myriad showed that the m圜hoice HRD test predicted drug response to platinum therapy in certain patients with triple-negative breast and ovarian cancers. Positive m圜hoice HRD scores, reflective of DNA repair deficiencies, are prevalent in all breast cancer subtypes, ovarian and most other major cancers. The m圜hoice HRD score is a composite of three proprietary technologies: loss of heterozygosity, telomeric allelic imbalance and large-scale state transitions. Myriad`s m圜hoice HRD is the most comprehensive homologous recombination deficiency test to detect when a tumor has lost the ability to repair double-stranded DNA breaks, resulting in increased susceptibility to DNA-damaging drugs such as platinum drugs or PARP inhibitors. The collaboration with TESARO began in March 2014 and includes several ongoing clinical trials in a variety of tumor types. The m圜hoice HRD test is being developed in parallel with the clinical development of niraparib. The key findings are illustrated in the chart below. The NOVA results showed that in patients who were germline BRCA mutation carriers, the median PFS for patients treated with niraparib was 21.0 months compared to 5.5 months for the control group (p "We estimate that m圜hoice HRD identifies more than double the number of patients who may benefit compared to germline BRCA testing alone." "Patients with ovarian cancer who tested positive with m圜hoice HRD experienced a clinically meaningful improvement in PFS," said Johnathan Lancaster, M.D., Ph.D., gynecologic oncologist and chief medical officer of Myriad Genetic Laboratories.
Hrd test trial#
This trial was designed to assess progression free survival (PFS) in a broad population of patients who were assigned to one of two cohorts based upon germline BRCA mutation status. NOVA is a well-controlled Phase 3 trial of niraparib that enrolled 553 patients with recurrent ovarian cancer who responded to their most recent platinum-based chemotherapy.
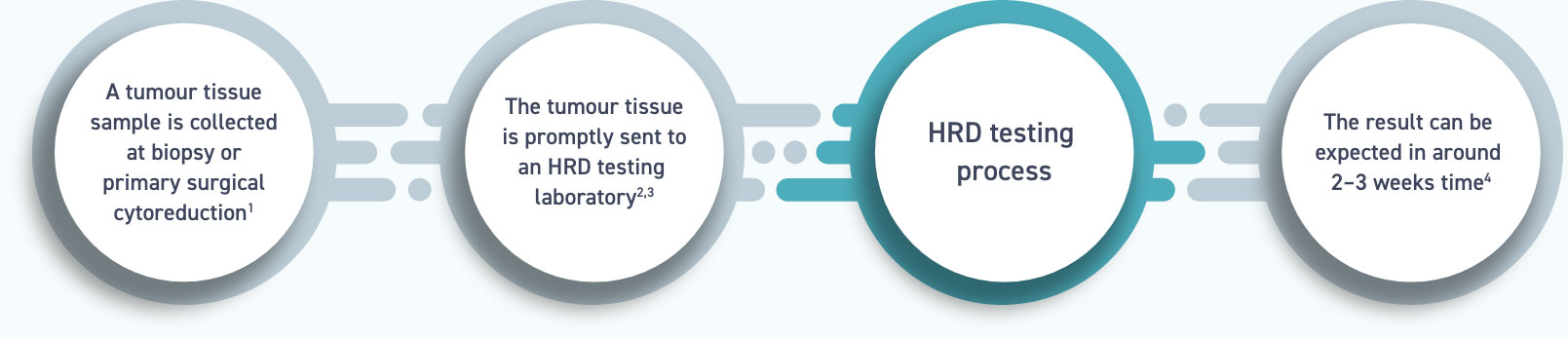
Today`s announcement follows publication of the NOVA study in the New England Journal of Medicine. The m圜hoice HRD test was evaluated in the NOVA study (NCT01847274) of nirarapib, an investigational oral PARP inhibitor being developed by TESARO ( TSRO). ( MYGN), a leader in molecular diagnostics and personalized medicine, today announced that its m圜hoice ® HRD test identified more than double the number of patients who may benefit from treatment with niraparib than were identified by germline BRCA testing alone. 08, 2016 (GLOBE NEWSWIRE) - Myriad Genetics, Inc.
